Ascend Trial, the screening
MS is a progressive autoimmune disease and so far no cure has been found. It is devastating; you just gradually lose the ability to control part or parts of your body.
Ascend trial, the screening
My left leg has no strength. generally speaking from the wast down I’m a bit of a disaster. At least I can walk a short distance. I now have secondary progressive MS, initially it was relapsing remitting MS.
People with relapsing remitting MS can be prescribed the drug Tysabri and this reduces the progression of MS significantly. For a few people it seems to completely stop the progression. The damage to the nerves and the myelin can not be repaired but things can change.
The ASCEND trial will investigate whether treatment with the Tysabri drug can prevent worsening in walking, hand movement and daily functioning of people with secondary progressive multiple sclerosis. Scientists say preliminary data shows Tysabri has the potential to reduce MS inflammation in the brain and “reduce secondary progressive MS-related disease progression.”
Is this why trials cost millions?
Around 850 patients in 15 countries will take part in the drug trial, including trial sites in the UK. Study participants will be aged between 18 – 58 years old with a diagnosis of secondary progressive MS for at least two years.
I stopped work in January 2012, medically retired. I no longer had the energy to haul myself, luggage and mobility scooter around Europe for contract work. My neurologist, Professor Giovannoni then suggested that I put myself forward for this trial, The selection process was strict; I was selected. I think I assumed that I would be selected so it would have worried me if I was dscarded.
Double blinded random trial

9 hole peg test
The screening included the following, measuring how far I could walk, standard 9 hole peg test, physical examination, measurement of my co-ordination. They needed blood samples and urine samples and the whole process took about 2 – 3 hours. I was not allowed to talk to the doctor. He was blinded so he knew nothing of my MS history, obviously I had never met him before.
What have I got to lose?
The screening process showed that I am PML positive (progressive multifocal leukoencephalopathy). All I will say is that it is inflamation of the brain, very nasty, can be fatal and affects about 1 in 500. Its a risk I am prepared to take. The trial is a double blind with half the people on a placebo so the chances are 1 in 1000. Put it this way, if I decide not to take the risk then my MS will definitely get worse and worse,. If I do go on the trial then there is a chance the the rate of progression of MS will be reduced. To my mind it was a ‘no brainer’ decision.
The initial screening was in late July and this included an MRI scan. There is then a 6 week window before the first infusion. So far so good but there was a fly in the ointment, I had to have a lumbar puncture. This is a process that allows a quantity of cerebrospinal fluid to be extracted from the spine.
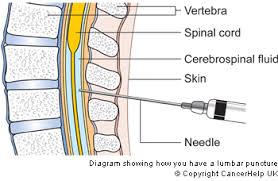
Lumbar puncture diagram at base of the spine – ouch
Lumbar puncture, not pleasant
It should not be a painful procedure but the idea of a needle being injected into my back was very stressful. I had to hold and squeeze the nurse’s hand because I could hear the doctor saying that the first attempt was unsuccessful. Ceri, the clinical research nurse, was a real hero, it was her hand that I squeezed. I was so tempted to get up and walk away. The procedure lasted over 45 minutes but it felt a lot, lot longer.
Paralympics
I had the lumbar puncture on Friday 31 August and afterwards I went to the Paralympics, down the road from the Royal London Hospital. It was a fantastic event and so well organised, I completely forgot about disability and MS while I was there.
Next time I will talk about the first infusion.
People with relapsing remitting MS can be prescribed the drug Tysabri and this reduces the progression of MS significantly. For a few people it seems to completely stop the progression. The damage to the nerves and the myelin cannot be repaired but things do change…
September 2012



“No brainer” appeals to me…
Hello David,
The words do have a certain appeal
Do you now know whether you were on a placebo? And did the Tysabri have any effect if you weren’t on the placebo? John
Hello John,
Never found out if I was on a placebo. Trial was canned cos Tysabri does not improve mobility/EDSS in SPMS, that is what they were testing. Much later it was realised that Tysabri helps to reduce loss of use of the upper limbs, ie hands and arms and ultimately the #ThinkHand project was set in place by the Barts MS team.